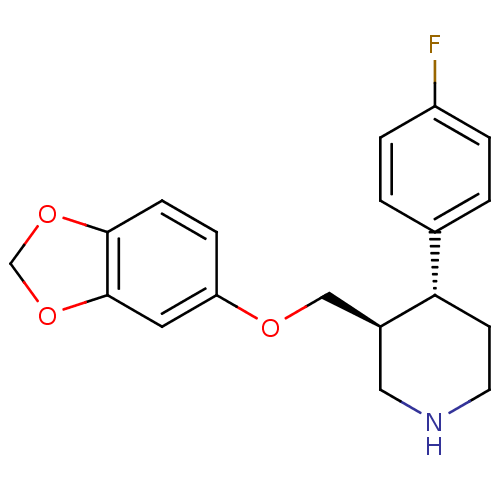
BDBM22416 (3S,4R)-3-[(1,3-benzodioxol-5-yloxy)methyl]-4-(4-fluorophenyl)piperidine::(3S,4R)-3-[(2H-1,3-benzodioxol-5-yloxy)methyl]-4-(4-fluorophenyl)piperidine::CHEMBL490::PAROXETINE::US09969700, Paroxetine::US9944618, Compound ID No. 182::[3H]Paroxetine
SMILES Fc1ccc(cc1)[C@@H]1CCNC[C@H]1COc1ccc2OCOc2c1
InChI Key InChIKey=AHOUBRCZNHFOSL-YOEHRIQHSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 22416
Found 5 hits for monomerid = 22416
TargetBeta-adrenergic receptor kinase 1(Homo sapiens (Human))
Hefei University Of Technology
Curated by ChEMBL
Hefei University Of Technology
Curated by ChEMBL
Affinity DataIC50: 1.10E+3nMAssay Description:Inhibition of GRK2 (unknown origin)More data for this Ligand-Target Pair
TargetBeta-adrenergic receptor kinase 1(Homo sapiens (Human))
Hefei University Of Technology
Curated by ChEMBL
Hefei University Of Technology
Curated by ChEMBL
Affinity DataIC50: 1.38E+3nMAssay Description:Binding affinity of compound towards dopamine transporter determined using [3H]WIN-35 428 as radioligandMore data for this Ligand-Target Pair
Affinity DataIC50: 1.38E+3nMAssay Description:Inhibition of bovine GRK2 S670A mutant after 5 mins in presence of ATP by phosphorimaging assayMore data for this Ligand-Target Pair
TargetBeta-adrenergic receptor kinase 1(Homo sapiens (Human))
Hefei University Of Technology
Curated by ChEMBL
Hefei University Of Technology
Curated by ChEMBL
Affinity DataKd: 2.98E+3nMAssay Description:Binding affinity to full length human GRK2 by Thermofluor thermal shift assayMore data for this Ligand-Target Pair
Affinity DataIC50: 780nMAssay Description:Inhibition of bovine GRK2 using porcine brain tubulin as substrate incubated for 3 to 5 mins by [gamma32P]ATP based radiometric assayMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)